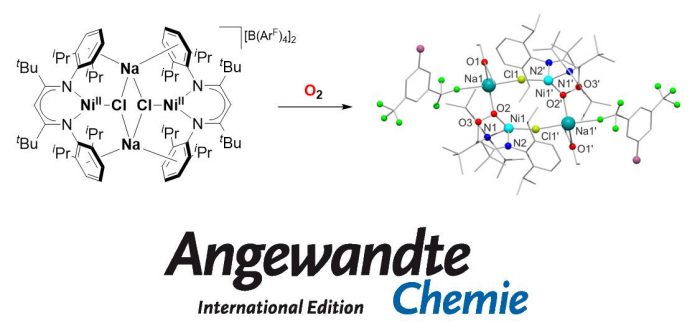
In metal-mediated O2 activation, nickel(II) compounds hardly play a role, but recently it has been shown that enzymes can use nickel(II) for O2 activation. Now a low-coordinate Lewis acidic nickel(II) complex has been synthesized that reacts with O2 to give a nickel(II) organoperoxide, as proposed for the enzymatic system. Its formation was studied further by UV/Vis absorption spectroscopy, leading to the observation of a short-lived intermediate that proved to be reactive in both oxygen atom transfer and hydrogen abstraction reactions, while the peroxide efficiently transfers O atoms. Both for the enzyme and for the functional model, the key to O2 activation is proposed to represent a concomitant electron shift from the substrate/co-ligand. The results were published today in Angewandte Chemie (Angew. Chem. Int. Ed. 2017, ASAP):
P. Holze, T. Corona, N. Frank, B. Braun-Cula, C. Herwig, A. Company, and C. Limberg
“Activation of Dioxygen at a Lewis Acidic Nickel(II) Complex: Characterization of a Metastable Organoperoxide Complex”
Angew. Chem. Int. Ed. 2017, [], ASAP-
[abstract]
DOI: 10.1002/anie.201609526
- sec.iqcc@udg.edu
- +34 972 41 83 57