Joint experimental/computational studies in chemistry provide new complementary insights which are unattainable by experimental or computational chemistry alone. This is confirmed once again by a recent paper in the Proceedings of the National Academy of Sciences USA by research groups of Prof. Que (Univ. Minnesota) and Prof. Swart (ICREA and Univ. Girona). They collaborated in understanding the origin of substantial rate enhancements observed in experiments with iron complexes based on the TMC (tetramethylcyclam) ligand.
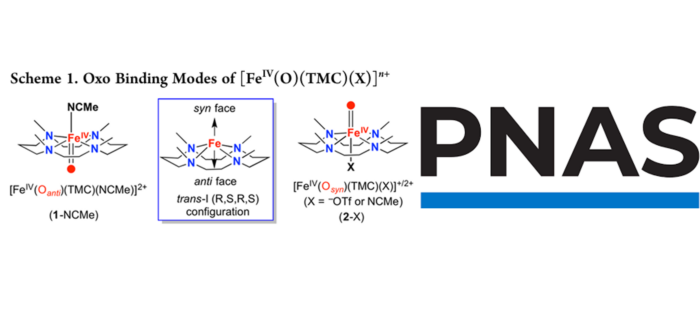
Twenty years ago, Prof. Que and co-workers reported the first crystallographic structure of a high-valent nonheme Fe(IV)-oxo complex,[1] which was characterized through spectroscopy to confirm the +4 oxidation state of iron. In this complex the oxo atom is found at the opposite side of the ligand from the methyl groups (TMC-anti, see Scheme 1, left). In 2015, they were able to synthetize the upside-down complex,[2] where the oxo atom is found on the same side as the methyl groups of the ligand (TMC-syn, Scheme 1, right). The effect of this orientation of the oxo group on reactivity had not been reported until recently.
High-valent iron complexes, e.g. the Fe(IV)-oxo examples above, are often involved in essential processes in metalloenzymes, like hydrogen-atom transfer (HAT) reactions and oxygen-atom transfer reactions (OAT), including olefin epoxidation. Experiments in Que’s lab in Minnesota show that TMC-syn reacts with HAT substrates at 1.3 to 3-fold faster rates than TMC-anti. Surprisingly, the reactivity difference increases dramatically in OAT reactions. R2S substrates are oxidized into R2S=O products at rates 2-to-3 orders of magnitude faster by TMC-syn than TMC-anti. Even more remarkably, TMC-syn epoxidizes all the olefin substrates in this study, while TMC-anti reacts only with cis-cyclooctene but at a 100-fold slower rate.
Comprehensive quantum chemical calculations in the Swart lab in Girona have uncovered the key factors that govern the intriguing reactivity differences between these two topological isomers. The iron-oxo unit moves out of the plane of the TMC ligand in TMC-syn, but not in TMC-anti, and does even more so as the reaction progresses. With OAT reactions the axial acetonitrile dissociates from iron, allowing the s* acceptor orbital to be stabilized and enhance reactivity. This orbital is not involved in HAT reactions, and hence no such effect of acetonitrile-dissociation (and accompanying rate enhancement) occurs.
In 2022, Prof. Que was elected as member of the prestigious National Academy of Sciences (NAS) of the US for his excellence in original scientific research.[3] Membership in the NAS is one of the highest honors given to a scientist or engineer. Newly elected NAS members are given the opportunity to present an inaugural contribution to the Proceedings of the National Academy of Sciences USA.
It has been recently published in Proc. Natl. Acad. Sci. USA:
B. Chandra, F. Ahsan, Y. Sheng, M. Swart, L. Que, Jr.
“A tale of two topological isomers: Uptuning [FeIV(O)(Me4cyclam)]2+ for olefin epoxidation”
Proc. Natl. Acad. Sci. USA 2024, ASAP
References
[1] J-U. Rohde, J-H. In, M.H. Lim, W.W. Brennessel, M.R. Bukowski, A. Stubna, E. Münck, W. Nam, L. Que Jr, Science 2003, 299, 1037-1039, DOI 10.1126/science.299.5609.1037
[2] J. Prakash, G.T. Rohde, K.K. Meier, E. Münck, L. Que, Jr, Inorg. Chem. 2015, 54, 11055–11057, DOI 10.1021/acs.inorgchem.5b02011
[3] https://cse.umn.edu/chem/news/professor-larry-que-elected-national-academy-sciences
Girona, March 14th, 2024
For more info: ges.iqcc@udg.edu