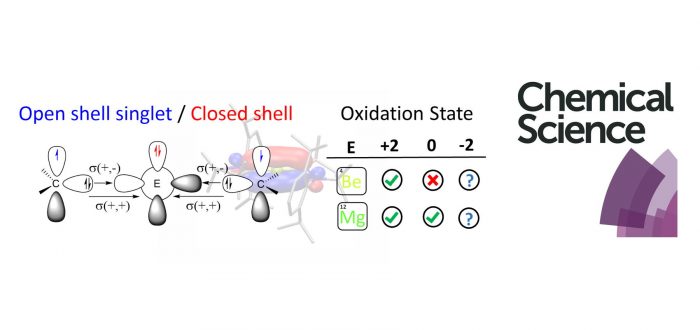
The synthesis and characterization of low-valent group 2 (E = Be and Mg) stabilized compounds have been long pursued. In this article we discuss the electronic structure of a series of Lewis base-stabilized Be and Mg compounds. Despite the accepted zero(0) oxidation state nature of the group 2 elements of some recent experimentally accomplished species, our chemical-bonding analysis performed on multireference (CASSCF) wavefunctions provides compelling evidence for a strong diradical(oid) character, and thus an oxidation state of +2. We elaborate on the distinction between a description as a donor-acceptor interaction L(0) <=> E(0) <=> L(0) and the internally oxidized situation, better interpreted as a diradicaloid L(1) => E(+2) <= L(1) species. The experimentally accomplished examples rely on the strengthened bonds by increasing the pi-acidity of the ligand; avoiding this interaction could lead to an unprecedented low-oxidation state.
This study is the result of an international cooperation between several research groups and mainly lead by the Andrada’s (Saarland university) and Salvador’s (Universitat de Girona) teams, and has recently been published in Chemical Science:
M. Gimferrer, S. Danés, E. Vos, C.B. Yildiz, I. Corral, A. Jana, P. Salvador and D.M. Andrada
“The oxidation state in low-valent beryllium and magnesium compounds”
Chem. Sci. 2022, 13, 6583-6591
DOI: 10.1039/D2SC01401G
Girona, June 6, 2022
For more info: gestor.iqcc@gmail.com