Oxygenation of organic substrates by iron metalloenzymes occurs with high levels of selectivity, mainly because of the exquisite control of both the primary and the secondary sphere of the iron center. Lots of efforts have been done in reproducing the efficiency of enzymatic systems, mainly tuning the first coordination sphere of the metal with very well-defined ligands.
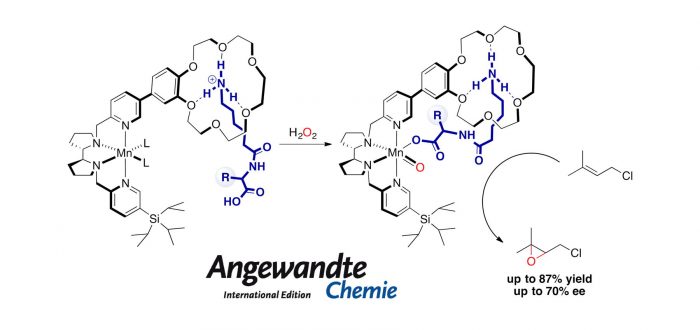
One of the most studied families are Fe and Mn complexes with tetradentate bis-amine-bis-pyridine ligands. These coordination compounds can efficiently activate hydrogen peroxide to form metal-oxo species able to oxidize a large range of substrates. Usually, a proton source is required for peroxide activation and specially with manganese systems, a high excess of carboxylic acid is required.
In this work, the authors use a supramolecular strategy for enzyme-like hydrogen peroxide activation at a manganese catalyst, using nearly stoichiometric amounts of carboxylic acid respect to the metal. This approach relies on supramolecular recognition of an alpha,omega-amino acid in the second coordination sphere of the metal to locate the carboxylic acid moiety in an optimal position to access the first coordination sphere and get involved in the catalytic cycle, resulting in an effective H2O2 activation, leading to catalytic asymmetric epoxidation.
The study was recently published in Angewandte Chemie International Edition:
L. Vicens, G. Olivo, and M. Costas
“Remote Amino Acid Recognition Enables Effective Hydrogen Peroxide Activation at a Manganese Oxidation Catalys”
Angew. Chem. Int. Ed. 2021, ASAP-
DOI: 10.1002/anie.202114932
Girona, Dec. 14, 2021
For more information: gestor.iqcc@gmail.com