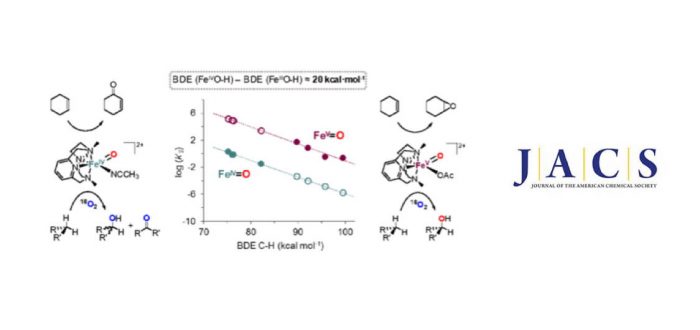
This work directly compares the spectroscopic and reactivity properties of an oxoiron(IV) and an oxoiron(V) complex that are supported by the same neutral tetradentate N-based PyNMe3 ligand. A complete spectroscopic characterization of the oxoiron(IV) species (2) reveals that this compound exists as a mixture of two isomers. The reactivity of the thermodynamically more stable oxoiron(IV) isomer (2b) is directly compared to that exhibited by the previously reported 1e–oxidized analogue [FeV(O)(PyNMe3)(OAc)]2+ (3). Our data indicates that 2b is 4 to 5 orders of magnitude slower than 3 in hydrogen atom transfer (HAT) from C-H bonds. The origin of this huge difference lies in the strength of the O-H bond formed after HAT by the oxoiron unit, the O-H bond derived from 3 being about 20 kcal·mol-1 stronger than that from 2b. The estimated bond strength of the FeIVO-H bond of 100 kcal·mol-1 is very close to the reported values for highly active synthetic models of compound I of cytochrome P450. In addition, this comparative study provides direct experimental evidence that the lifetime of the carbon-centered radical that forms after the initial HAT by the high valent oxoiron complex depends on the oxidation state of the nascent Fe-OH complex. Complex 2b generates long-lived carbon-centered radicals that freely diffuse in solution, while 3 generates short-lived caged radicals that rapidly form product C-OH bonds, so only 3 engages in stereoretentive hydroxylation reactions. Thus, the oxidation state of the iron center not only modulates the rate of HAT but also the rate of ligand rebound. It was published recently in Journal of the American Chemical Society.
V. Dantignana, J. Serrano-Plana, A. Draksharapu, C. Magallón, S. Banerjee, R. Fan, I. Gamba, Y. Guo, L. Que, M. Costas, and A. Company
“Spectroscopic and Reactivity Comparisons between Nonheme Oxoiron(IV) and Oxoiron(V) Species Bearing the Same Ancillary Ligand”
J. Am. Chem. Soc. 2019, 141, 15078-15091 [abstract]
DOI: 10.1021/jacs.9b05758