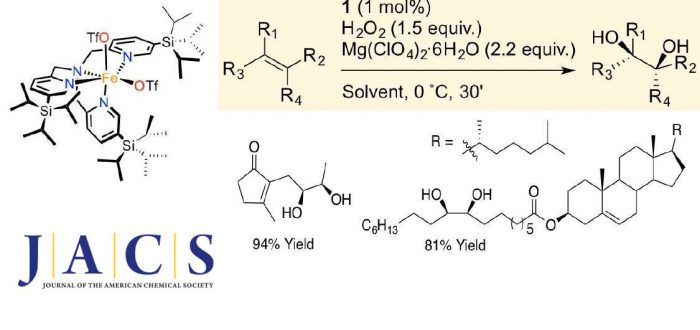
Product release is the rate determining step in the arene syn-dihydroxylation reaction taking place at Rieske oxygenase enzymes, and is regarded as a difficult problem to be resolved in the design of iron catalysts for olefin syn-dihydroxylation with potential utility in organic synthesis. Towards this end, in this work a novel catalyst bearing a sterically encumbered tetradentate ligand based in the tpa (tpa = tris-(2-methylpyridyl)amine scaffold, [FeII(CF3SO3)2(5-tips3tpa)], 1 has been designed. The steric demand of the ligand was envisioned as a key element to support a high catalytic activity by isolating the metal center, preventing bimolecular decomposition paths, and facilitating product release. In synergistic combination with a Lewis acid that helps sequestering the product, 1 provides good to excellent yields of syn-diol products (up to 97% isolated yield), in short reaction times under mild experimental conditions using a slight excess (1.5 equiv.) of aqueous hydrogen peroxide, from the oxidation of a broad range of olefins. Predictable site selective cis-dihydroxylation of diolefins is shown. The encumbered nature of the ligand also provides a unique tool that has been used in combination with isotopic analysis to define the nature of the active species and the mechanism of activation of H2O2. Furthermore 1 is shown to be a competent synthetic tool for preparing O-labelled diols using water as oxygen source.
The results were published today in Journal of the American Chemical Society (J. Am. Chem. Soc. 2017, ASAP):
M. Borrell, and M. Costas
“Mechanistically driven development of an iron catalyst for selective syn-dihydroxylation of alkenes with aqueous hydrogen peroxide”
J. Am. Chem. Soc. 2017, [], ASAP-
[abstract]
DOI: 10.1021/jacs.7b07909