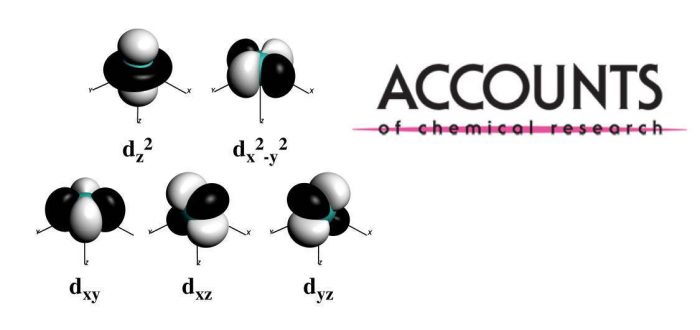
Spin states of transition-metal complexes are essential for understanding many cases in biology, medicine, catalysis, and photonics. The choice of theoretical method, basis set, solvation and many other effects represent factors that need to be considered to be able to deal with the close-lying spin states in coordination compounds. However, the assignment of spin states is not unambiguous. A combination of synthesis, theoretical simulations, crystallographic and spectroscopic characterization has to be used to tackle the problems at hand, which has led to enrichment of our understanding of transition-metal chemistry and new approaches such as Shaik’s exchange-enhanced reactivity, Baerends’ low-lying acceptor orbitals or Mayer’s driving force. In seeking to develop appropriate DFAs for the treatment of spin states and weak interactions, it became clear that there is an intriguing link between the spin states of transition metals and SN2 barriers of organic molecules.
Our work has uncovered multiple examples of the influence of the spin state on the coordination environment of a transition metal. It demonstrated the possibility to question the experimentally predicted spin and oxidation states, and give rationalization for the nature of the spin state preferences in important bio-mimetic species. Most importantly, through detailed studies on these systems we have begun to develop and provide key insights into the subtle interplay between the complicated electronic structure with the structural, mechanistic and spectroscopic observables.
The results were published today in Accounts of Chemical Research:
M. Swart and M. Gruden
“Spinning around in transition-metal chemistry”
Acc. Chem. Res. 2016, ASAP
[abstract] #OpenAccess
DOI: 10.1021/acs.accounts.6b00271