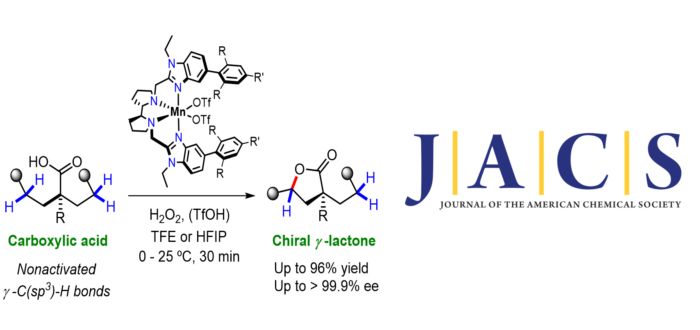
Chiral oxygenated aliphatic moieties are recurrent in biological and pharmaceutically relevant molecules, and constitute one of the most versatile type of functionalities for further elaboration. In this work, Arnau Call, Giorgio Capocasa, Andrea Palone, Laia Vicens, Eric Aparicio, Najoua Choukairi Afailal, Nikos Siakavaras, Maria Eugènia López Saló, Massimo Bietti and Miquel Costas report a protocol for straightforward and general access to chiral ?-lactones via enantioselective oxidation of strong nonactivated primary and secondary C(sp3)-H bonds in readily available carboxylic acids. The method reported here provides the straightforward conversion of cheap and simple carboxylic acids into highly added-value chiral compounds with unprecedented levels of enantioselectivity.
The key enabling aspect is the use of robust sterically encumbered manganese catalysts that provide outstanding enantioselectivities (up to >99.9%) and yields (up to 96%). The use of catalysts based on an earth-abundant metal singularizes the current system with respect to asymmetric C(sp3)?H oxidation reactions that rely on precious metals. In addition, the use of hydrogen peroxide as an oxidant confers to the system a high atom economy, which combined with the mild experimental conditions makes the reaction particularly appealing from sustainability and large-scale applicability perspectives. The simplicity of the reaction, composed by low loading of a manganese catalyst and hydrogen peroxide in a fluorinated alcohol solvent at ambient temperature, contrasts with the state-of-the-art systems constituted by multiple reagents and additives often accompanied by prolonged heating at high temperatures, producing large amounts of waste.
The availability of the substrates and the immediate importance of the resulting products in natural product synthesis and materials combined with their facile follow-up elaboration constitute a distinctive aspect of the current reactions. We envision that the high structural versatility and powerful oxidation ability of the novel family of catalysts disclosed in this work will find utility in the development of novel enantioselective C?H functionalization reactions.
This work has been performed in collaboration of Prof. Massimo Bietti (Università “Tor Vergata”, Italy), and Dr. Arnau Call, Giorgio Capocasa, Andrea Palone, Dr. Laia Vicens, Eric Aparicio, Najoua Choukairi, Nikos Siakavaras, Maria Eugènia López, Prof. Miquel Costas of the QBIS-CAT group of the Institute of Computational Chemistry and Catalysis (IQCC) of the University of Girona.
It has been recently published open access in Journal of the American Chemical Society:
A. Call,* G. Capocasa, A. Palone, L. Vicens, E. Aparicio, N. Choukairi Afailal, N. Siakavaras, M. E. López Saló, M. Bietti,* and M. Costas*.
“Highly enantioselective catalytic lactonization at nonactivated primary and secondary g-C-H bonds”
J. Am. Chem. Soc. 2023, 145, 18094?18103
DOI: 10.1021/jacs.3c06231
Girona, September 27, 2023
For more info: ges.iqcc@udg.edu