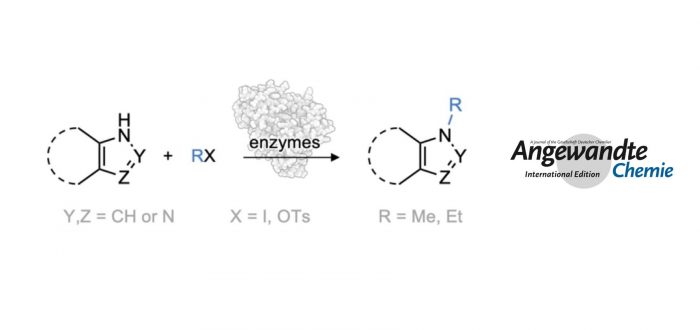
Methods for regioselective N-methylation and -alkylation of unsaturated heterocycles with “off the shelf” reagents are highly sought-after. This reaction could drastically simplify synthesis of privileged bioactive molecules. Here we report engineered and natural methyltransferases for challenging N-(m)ethylation of heterocycles, including benzimidazoles, benzotriazoles, imidazoles and indazoles. The reactions are performed through a cyclic enzyme cascade that consists of two methyltransferases using only iodoalkanes or methyl tosylate as simple reagents. This method enables the selective synthesis of important molecules that are otherwise difficult to access, proceeds with high regioselectivity (r.r. up to >99%), yield (up to 99%), on a preparative scale, and with nearly equimolar concentration of simple starting materials.
This project was led by Jun.-Prof. Stephan Hammer (Univ. Bielefeld, Germany) and his Team. Dr. Garcia-Borràs, PI at the IQCC, and his PhD student Jordi Soler collaborated to study the molecular basis for the promiscuity exhibited by these methyl transferases using computational modelling.
The results of this project have been recently published in Angewandte Chemie:
F. Ospina, K. H. Schülke, J. Soler, A. Klein, B. Prosenc, M. Garcia-Borràs, and S. C. Hammer
“Selective Biocatalytic N-Methylation of Unsaturated Heterocycles”
Angew. Chem. Int. Ed., 2022, ASAP
DOI: 10.1002/anie.202213056
Girona, Oct. 19, 2022
For more info: gestor.iqcc@gmail.com