A recent paper (Kim et al. Nature Commun. 2019, 10, 4983) claims that there is a double charge transfer in the first singlet excited singlet state in TMTQ (an oligomer composed of a central 1,6-methano[10]annulenes, M10A, and 5-diacyanomethyl-thiophene exocyclic groups, see Figure 1) that is stabilized by the Baird aromaticity acquired by the central M10A ring.
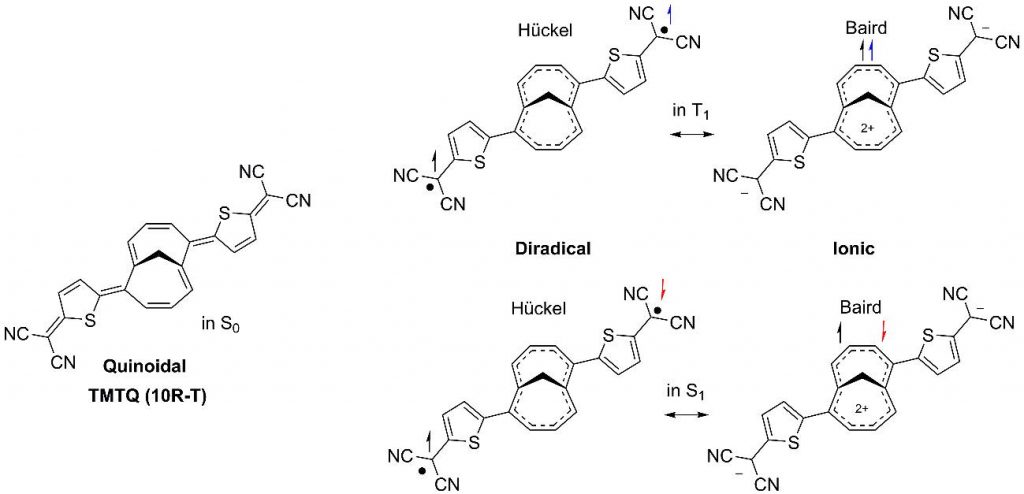
In a previous work (Jorner et al. Chem. Eur. J. 2016, 22, 2793), some of us found that the charge transfer in the first excited singlet and triplet states in TMTQ is minor and that the central ring in these low-lying excited states is a Hückel-Baird hybrid with a ca. 85% of Hückel and 15% of Baird character. In the light of the new existing experimental data, Claire Tonnelé and Prof. Henrik Ottosson of the Uppsala University, Dr. David Casanova and Dr. Eduard Matito of the Donostia International Physics Center (previous DiMoCat member), and Sílvia Escayola, Dr. Albert Poater, and Prof. Miquel Solà of the IQCC of the University of Girona decided to reinvestigate the problem using (time-dependent) density functional theory methods. We considered not only TMTQ but also a series of symmetrically substituted conjugated rings that can generate Baird aromaticity in the lowest-lying excited states. This work shows that the interpretation by Kim et al. needs to be revised because low-lying excitations of symmetrically substituted conjugated rings including TMTQ hold very weak charge transfer character. Our computational results also allow us to establish general guidelines for the rational design of molecules with excited state Hückel/Baird aromaticity in pro-aromatic quinoidal compounds We found that high Baird character of the central ring is achieved only with anionic and small conjugated central rings with electron donating groups as substituents and small exocyclic groups with electron withdrawing substituents. Our study aims to warn about the need to make correct interpretations of the experiments in this growing area of research, as only then can the excited state aromaticity concept develop in a useful tool for the design of high-performance organic electronic devices.
The paper was published recently in Angewandte Chemie:
S. Escayola, C. Tonnelé, E. Matito, A. Poater, H. Ottosson, M. Solà, and D. Casanova
“Guidelines for Tuning the Excited State Hückel?Baird Hybrid Aromatic Character of Pro?Aromatic Quinoidal Compounds”
Angew. Chem. Int. Ed. 2021, ASAP-
DOI: 10.1002/anie.202100261
Girona, Feb. 15, 2021
For more info: gestor.iqcc@gmail.com