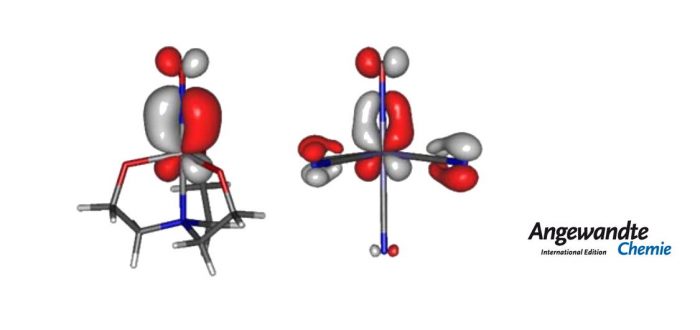
The nitrosyl ligand is the prototypic example of a so-called ‘non-innocent’ ligand, which hinders the unambiguous assignment of the central metal’s oxidation state (OS). Even after the IUPAC effort to revise the concept of OS, ambiguities still originate on the M–N–O angle, where it is widely assumed the rule “the MNO segment should be linear for NO+ but bent for NO?”. Missing a clear-cut guideline, the nitrosylmetal community has continued to apply workarounds for the assignment of OSs.
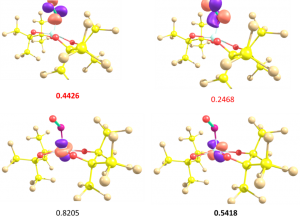
In this work we have made use of the effective-oxidation-state (EOS) method, introduced by Pedro Salvador and coworkers at the Universitat de Girona for the study and categorization of three isostructural nitrosylmetallates which were formed as adducts of nitric oxide to a special class of low-coordinate bis(diolato)metallates, the tetracoordinate bis(perfluoro pinacolato)metallates [M(fpin)2]2? of chromium(II), iron(II), and cobalt(II), synthesized and characterized by the group of Prof. Klüfers at the Ludwigs-Maximilians-Universität of München. Wieghardt’s diamagnetic [V(NO)(tea)]? ion has also been reconsidered in the light of EOS analysis, leading to a new assignment of the OS of the central V atom. Moreover, it has been realized that spin polarization of the M-N-O bonds is a key factor on the assignation of OS.
The study has been published recently in Angewandte Chemie:
T. Ampßler, G. Monsch, J. Popp, T. Riggenmann, P. Salvador, D. Schröder, and P. Kluefers
“Not Guilty on Every Count: the ‘Non?Innocent’ Nitrosyl Ligand in the Framework of IUPAC’s Oxidation?State Formalism”
Angew. Chem. Int. Ed. 2020, [], ASAP-
DOI: 10.1002/anie.202003122