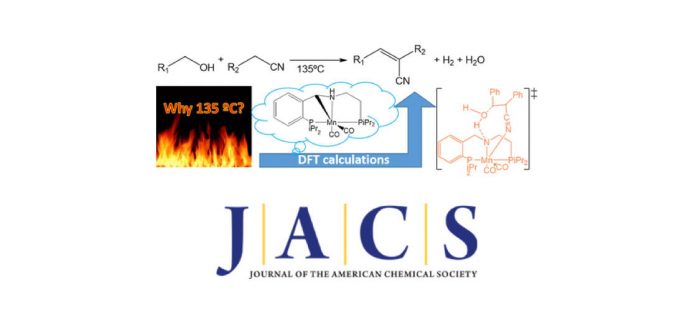
A pure computational paper by Miquel Solà, Albert Poater and co-workers has been undertaken to favour the generation of H2, once known experimentally by Milstein and coworkers that Mn-pincer could catalyze the acceptorless dehydrogenative coupling of nitriles and alcohols to yield acrylonitriles. The reaction mechanism proposed in that work contained some intermediates that, in most of the cases, were not characterized. Moreover, one of the intermediates involved a charged separation, which is unlikely in apolar solvents. To clarify the reaction mechanism of this critical reaction, a DFT study was performed. Results prove the existence of a cooperative effect of the metal and the ligand in several steps of the catalytic cycle. It is unveiled the presence of several equilibria between isomeric intermediates where water, or the same alcohol reagent, takes part in assisting the proton transfer. Furthermore, the charge-separated structure proposed experimentally turned out to be a nearly pure covalent bond between the two expected charged moieties. More importantly catalytically speaking, the Knoevenagel condensation step that generates the acrylonitrile is found to be the rate-determining step. The paper was published today in the Journal of the American Chemical Society:
A. Luque-Urrutia, M. Solà, D. Milstein, and A. Poater
“Mechanism of the Manganese-Pincer Catalyzed Acceptorless Dehydrogenative Coupling of Nitriles and Alcohols”
J. Am. Chem. Soc. 2019, ASAP
DOI: 10.1021/jacs.8b11308