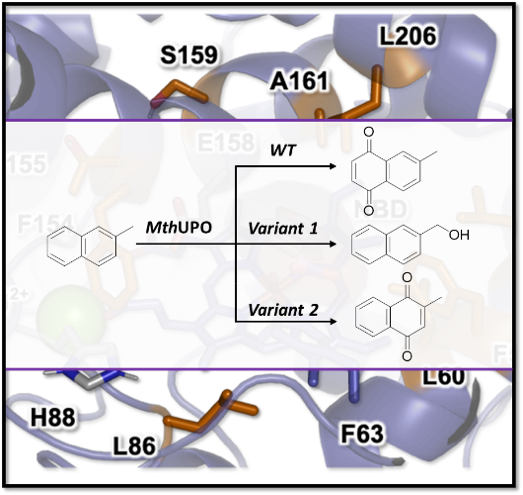
Unspecific peroxygenases (UPOs) enable oxyfunctionalizations of a broad substrate range with unparalleled activities. Tailoring these enzymes for chemo- and regioselective transformations represents a grand challenge due to the difficulties in their heterologous productions and lack of structural data. In a new collaboration by the research teams leaded by Dr. Martin Weissenborn (Leibniz Institute of Plant Biochemistry, Germany) and Dr. Marc Garcia-Borràs (IQCC), a new UPO from Myceliophthora thermophila, MthUPO, has been characterized and conducted to semi-rational protein engineering to enable new selective oxyfunctionalizations of organic substrates. PhD students Anja Knorrscheidt (from Leibniz Institute of Plant Biochemistry, Germany) and Jordi Soler (IQCC) have been in charge of the experiments and computations, respectively.
This protein engineering led to a significant reshaping of the active site as rationalized by extensive computational modelling carried out by Jordi Soler (PhD student working with Dr. Garcia-Borràs). The reshaping was responsible for the increased oxyfunctionalization activity, with improved kcat/Km values of up to 16.5-fold for the model substrate 5-nitro-1,3-benzodioxole. Moreover, variants were identified with high chemo- and regioselectivities in the oxyfunctionalization of aromatic and benzylic carbons, respectively.
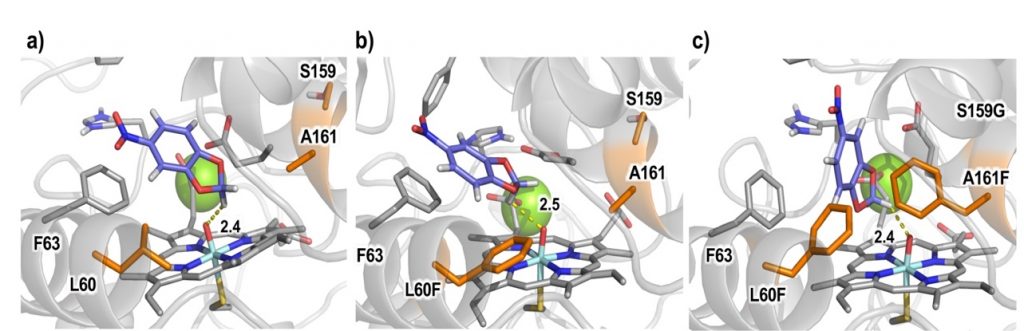
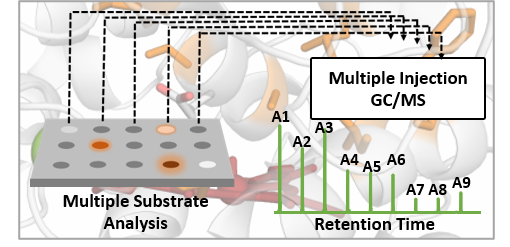
Following this line, a high throughput GC-MS approach was developed in the Weissenborn’s lab, permitting the simultaneous analysis of up to three substrates and six products quantitatively from one reaction mixture. This screening approach was applied to rationally build site-saturation libraries of the novel unspecific peroxygenase MthUPO. Using this setup enabled substantial insights from a relatively small mutant library: enzyme variants were identified exhibiting selective alkene epoxidation and substantially shifted regioselectivities to 2- and 1-octanol formations. Then, computational modelling was extensively applied by Jordi Soler to understand how these specific mutations modify and enhance the activities and selectivities for those particular substrates, providing important information that unravels the molecular basis by which the new enzyme variants are controlling these oxidation reactions.
The new proposed evolutionary protocol and rationalization of the enhanced activities and selectivities acquired by MthUPO variants represent a step forward toward the use and implementation of UPOs in biocatalytic synthetic pathways of industrial interest.
These results have been recently published in ACS Catalysis and Catalysis Science & Technology:
A. Knorrscheidt, J. Soler, N. Hünecke, P. Püllmann, M. Garcia-Borràs, and M.J. Weissenborn
“Accessing Chemo- and Regioselective Benzylic and Aromatic Oxidations by Protein Engineering of an Unspecific Peroxygenase”
ACS Catal. 2021, 11, 7327-7338
DOI: 10.1021/acscatal.1c00847 (Open Access Preprint)
A. Knorrscheidt, J. Soler, N. Hünecke, P. Püllmann, M. Garcia-Borràs, and M.J. Weissenborn
“Simultaneous screening of multiple substrates with an unspecific peroxygenase enabled modified alkane and alkene oxyfunctionalisations”
Catal. Sci. Technol. 2021, [], ASAP-
DOI: 10.1039/d0cy02457k (Open Access)
Girona, June 19, 2021
For more info: gestor.iqcc@gmail.com