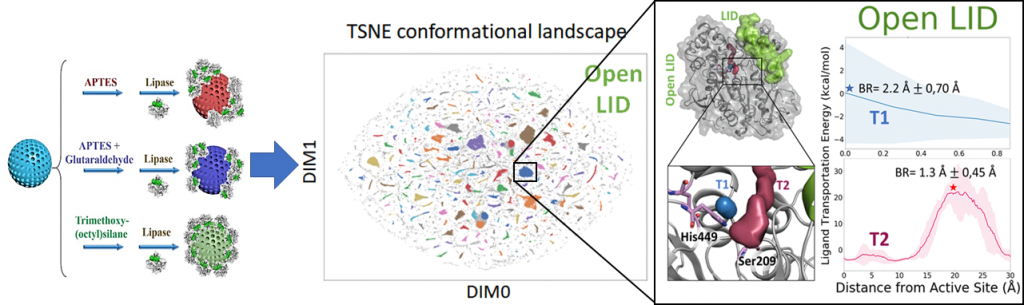
Enzyme powered micro/nanomotors have great potential applications in various areas, especially in the biomedical and environmental fields. To efficiently reach those applications, it is necessary to understand the fundamental aspects affecting the motion dynamics. In this collaboration project between IBEC (Prof. Sanchez), UCM (Dr. Filice), and IQCC (M. Estévez, Dr. Iglesias-Fernández, Prof. Osuna), we explored the impact of enzyme orientation on the performance of lipase-powered nanomotors by tuning the lipase immobilization strategies. The influence of the lipase orientation and lid conformation on substrate binding and catalysis was analyzed via Molecular Dynamics simulations coupled to dimensionality reduction techniques and CAVERdock simulations. The motion performance of the lipase-powered nanomotors indicates that the hydrophobic binding (via OTES) represents the best orienting strategy. In these conditions the lipase is immobilized with the lid domain in an open conformational state allowing substrate binding and catalysis. This leads to enhanced propulsion of the nanoparticle.
This work provides critical evidences of immobilization strategy and corresponding enzyme orientation for catalytic activity and in turn, the motion performance of nanomotors, thus helpful to future applications.
The paper was recently published in Angewandte Chemie:
L. Wang, M. Marciello, M. Estévez-Gay, P.E. Rodriguez, Y.L. Morato, J. Iglesias-Fernández, X. Huang, S. Osuna, M. Filice, and S. Sanchez
“Enzyme Conformation Influences the Performance of Lipase?powered Nanomotors”
Angew. Chem. Int. Ed. 2020, ASAP
DOI: 10.1002/anie.202008339
Girona, August 14, 2020
For more information: gestor.iqcc@gmail.com

