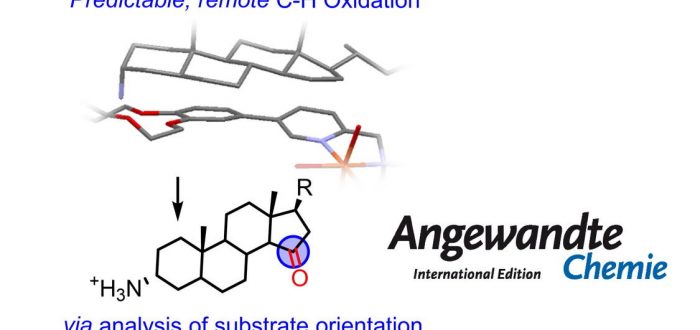
Predictability is a key requirement to encompass late-stage C-H functionalization in synthetic routes. However, prediction (and control) of reaction selectivity is usually challenging, especially for complex substrate structures and elusive transformations such as remote C( sp 3 )-H oxidation, as it requires to distinguish a specific C-H bond from many others with similar reactivity. Herein, we develop a strategy for predictable, remote C-H oxidation that entails substrate binding to a supramolecular Mn or Fe catalyst followed by elucidation of the conformation of the host-guest adduct via NMR analysis or, more easily, docking of crystal structures. These analyses indicate which remote C-H bonds are suitably oriented for the oxidation before carrying out the reaction, enabling prediction of site-selectivity. We applied this strategy to late-stage C( sp 3 )-H oxidation of aminosteroids at C15 (or C16) positions, with a selectivity tunable by modification of catalyst chirality and metal. The paper by Giorgio Olivo, Miquel Costas and co-workers appeared online recently in Angewandte Chemie-International Edition.
M. Costas, G. Olivo, G. Capocasa, B. Ticconi, O. Lanzalunga, and S. Di Stefano
“Predictable selectivity in remote C-H Oxidation of steroids via analysis of substrate binding mode”
Angew. Chem. Int. Ed. 2020, ASAP-
DOI: 10.1002/anie.202003078