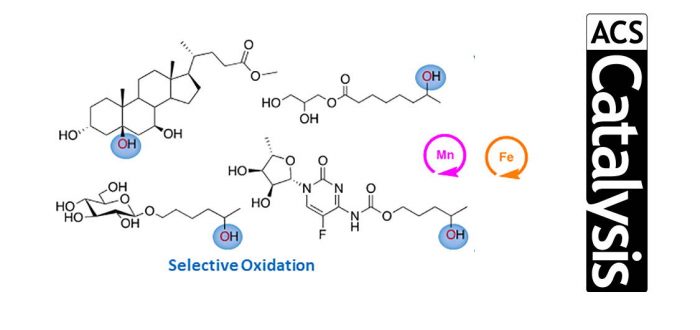
Site-selective and product chemoselective aliphatic C–H bond oxidation of 1,2-diols and of polyhydroxylated substrates using iron and manganese catalysts and hydrogen peroxide as terminal oxidant is described. The reaction capitalizes on the use of fluorinated alcohol solvents such as 2,2,2-trifluoroethanol (TFE) and 1,1,1,3,3,3-hexafluoro-2-propanol (HFIP), which exert a strong polarity reversal in the hydroxyl moieties of 1,2-diols via hydrogen bonding, in turn translating into a strong deactivation of proximal C–H bonds against a HAT initiated oxidation by the putative high-valent and electrophilic metal-oxo species. As a result, site-selective and product chemoselective oxidation of complex polyfunctional molecules such as steroids, sugars, and pharmaceuticals is described, where exclusive or predominant C–H bond hydroxylation at a remote and nonactivated site takes place. The current report discloses HAT initiated hydroxylations in fluorinated alcohol solvents as methods displaying orthogonal chemoselectivity to contemporary alcohol oxidations providing a useful tool for synthetic planning in densely functionalized molecules.
The paper was published recently in ACS Catalysis:
M. Borrell, S. Gil-Caballero, M. Bietti, and M. Costas
“Site-Selective and Product Chemoselective Aliphatic C–H Bond Hydroxylation of Polyhydroxylated Substrates”
ACS Catal. 2020, 10, 4702-4709
DOI: 10.1021/acscatal.9b05423