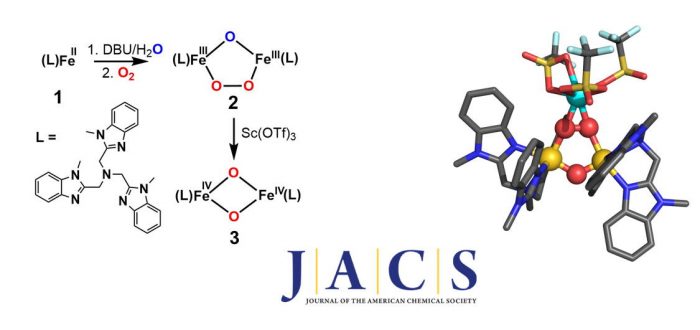
Soluble methane monooxygenase (sMMO) carries out methane oxidation at 4 °C and under ambient pressure in a catalytic cycle involving the formation of a peroxodiiron(III) intermediate (P) from the oxygenation of the diiron(II) enzyme and its subsequent conversion to Q, the diiron(IV) oxidant that hydroxylates methane. Synthetic diiron(IV) complexes that can serve as models for Q are rare and have not been generated by a reaction sequence analogous to that of sMMO. In this work, we show that [FeII(Me3NTB)(CH3CN)](CF3SO3)2(Me3NTB = tris((1-methyl-1H-benzo[d]imidazol-2-yl)methyl)amine) (1) reacts with O2 in the presence of base, generating a (mu-1,2-peroxo)diiron(III) adduct with a low O–O stretching frequency of 825 cm-1 and a short Fe···Fe distance of 3.07 Å. Even more interesting is the observation that the peroxodiiron(III) complex undergoes O–O bond cleavage upon treatment with the Lewis acid Sc3+ and transforms into a bis(mu-oxo)diiron(IV) complex, thus providing a synthetic precedent for the analogous conversion of P to Q in the catalytic cycle of sMMO.
The paper was recently published in the Journal of the American Chemical Society:
S. Banerjee, A. Draksharapu, P. Crossland, R. Fan, Y. Guo, M. Swart, and L. Que
“Sc3+-promoted O–O bond cleavage of a (?-1,2-peroxo)diiron(III) species formed from an iron(II) precursor and O2 to generate a complex with an FeIV2(?-O)2 core”
J. Am. Chem. Soc. 2020, ASAP [abstract]
DOI: 10.1021/jacs.9b12081