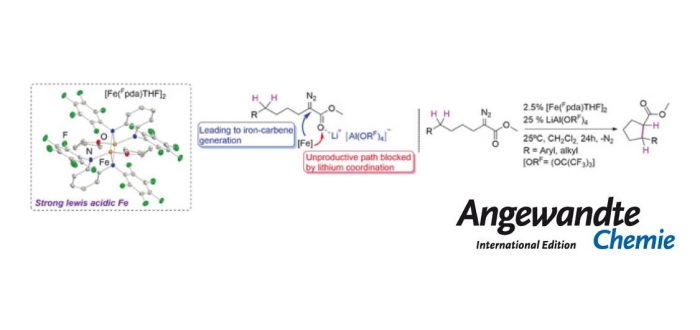
Combining an electrophilic iron complex [Fe(Fpda)(THF)]2 (3) [Fpda=N,N’-bis(pentafluorophenyl)-o-phenylenediamide] with the pre-activation of a-alkyl-substituted a-diazoesters reagents by LiAl(ORF)4 [ORF=(OC(CF3)3] provides unprecedented access to selective iron-catalyzed intramolecular functionalization of strong alkyl C(sp3)-H bonds. Reactions occur at 25°C via a-alkyl-metallocarbene intermediates, and with activity/selectivity levels similar to those of rhodium carboxylate catalysts. Mechanistic investigations reveal a crucial role of the lithium cation in the rate-determining formation of the electrophilic iron-carbene intermediate, which then proceeds by concerted insertion into the C-H bond. It was published recently in Angewandte Chemie-International Edition.
A. Hernán-Gómez, M. Rodríguez, T. Parella, and M. Costas
“Electrophilic Iron Catalyst Paired with a Lithium Cation Enables Selective Functionalization of Non-Activated Aliphatic C-H Bonds via Metallocarbene Intermediates”
Angew. Chem. Int. Ed. 2019, 58, 13904-13911 [abstract]
DOI: 10.1002/anie.201905986