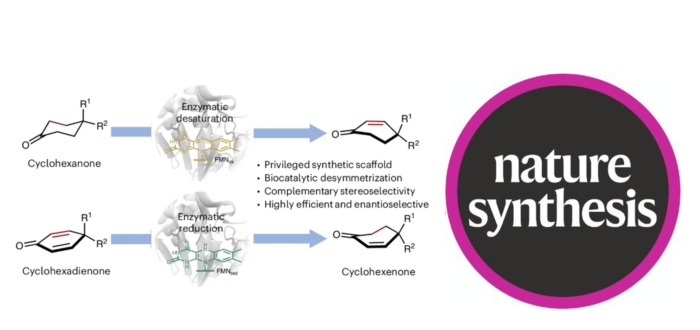
Efficient methods for achieving desaturation of carbonyl compounds are highly sought after in organic chemistry. In contrast to synthetic approaches, enzymatic desaturation systems offer the potential to enhance sustainability and selectivity but have remained elusive. Here we report the development of an enzymatic desaturation system based on flavin-dependent ene-reductases for desymmetrizing cyclohexanones. This platform facilitates the synthesis of a wide array of chiral cyclohexenones bearing quaternary stereocentres—structural motifs commonly present in bioactive molecules—with excellent yields and enantioselectivities. Experimental and computational mechanistic studies reveal the roles of key active-site residues that enable the formation and stabilization of an enolate intermediate in the desaturation event. Additionally, by leveraging these insights, we have devised a biocatalytic strategy for the synthesis of enones by reductively desymmetrizing cyclohexadienones. This method yields the opposite enantiomer compared to our desaturation system, underscoring the enantiodivergence and broad applicability of our flavin-based desymmetrization approaches.
TThis work has been carried out in collaboration between the group of Prof. Zhen Liu from the National Institute of Biological Sciences (China) and the group of Dr. Marc Garcia-Borràs from the IQCC. Dr. Carla Calvó-Tusell, a former PhD student from Marc’s group, was the leading computational author of the study.
It has been recently published open access in Nature Synthesis:
Q-Q Zeng, Q-Y Zhou, C. Calvó-Tusell, S.Y Dai, X. Zhao, M. Garcia-Borràs*, Z. Liu*(‡: These authors have equally contributed)
“Biocatalytic desymmetrization for synthesis of chiral enones using flavoenzymes”
Nat. Synth., 2024, 986.
DOI: 10.1038/s44160-024-00507-7
Girona, July 22nd, 2024
For more info: ges.iqcc@udg.edu