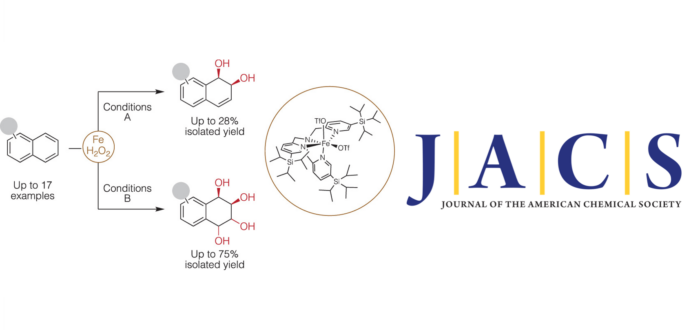
Arenes are interesting feedstocks for organic synthesis because of their natural abundance. However, the stability conferred by aromaticity severely limits their reactivity, mostly to reactions where aromaticity is retained. Methods for oxidative dearomatization of unactivated arenes are exceedingly rare but particularly valuable because the introduction of Csp3–O bonds transforms the flat aromatic ring in 3D skeletons and confers the oxygenated molecules with a very rich chemistry suitable for diversification. Mimicking the activity of naphthalene dioxygenase (NDO), a non-heme iron-dependent bacterial enzyme, herein we describe the catalytic syn-dihydroxylation of naphthalenes with hydrogen peroxide, employing a sterically encumbered and exceedingly reactive yet chemoselective iron catalyst. The high electrophilicity of hypervalent iron oxo species is devised as a key to enabling overcoming the aromatically promoted kinetic stability. Interestingly, the first dihydroxylation of the arene renders a reactive olefinic site ready for further dihydroxylation. Sequential bis-dihydroxylation of a broad range of naphthalenes provides valuable tetrahydroxylated products in preparative yields, amenable for rapid diversification.
It has been recently published open access in Journal of the American Chemical Society:
N. Choukairi Afailal, M. Borrell, M. Cianfanelli, and M. Costas*
“Dearomative syn-Dihydroxylation of Naphthalenes with a Biomimetic Iron Catalyst“
J. Am. Chem. Soc., 2023, ASAP
DOI: 10.1021/jacs.3c08565
Girona, January 9, 2024
For more info: ges.iqcc@udg.edu