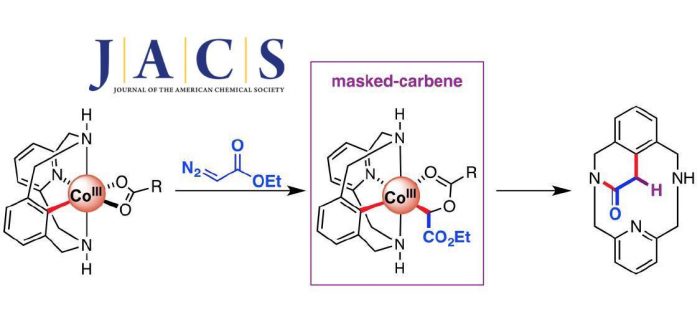
Herein we describe the synthesis of a family of aryl-Co(III)-carboxylate complexes and their reactivity with ethyl diazoacetate. Crystallographic, full spectroscopic characterization, and theoretical evidence of unique C-metalated aryl-Co(III) enolate intermediates is provided, unraveling a carboxylate-assisted formation of aryl-Co(III) masked-carbenes. Moreover, additional evidence for an unprecedented Co(III)-mediated intramolecular SN2-type C–C bond formation in which the carboxylate moiety acts as a relay is disclosed. This novel strategy is key to tame the hot reactivity of a metastable Co(III)-carbene and elicit C–C coupling products in a productive manner.
The results were published today in Journal of the American Chemical Society (http://www.dx.doi.org/10.1021/jacs.7b07880):
O. Planas, S. Roldán-Gómez, V. Martin-Diaconescu, T. Parella, J.M. Luis, A. Company, and X. Ribas
“Carboxylate-Assisted Formation of Aryl-Co(III) Masked-Carbenes in Cobalt-Catalyzed C–H Functionalization with Diazo Esters”
J. Am. Chem. Soc. 2017, [], ASAP- [abstract]
DOI: 10.1021/jacs.7b07880