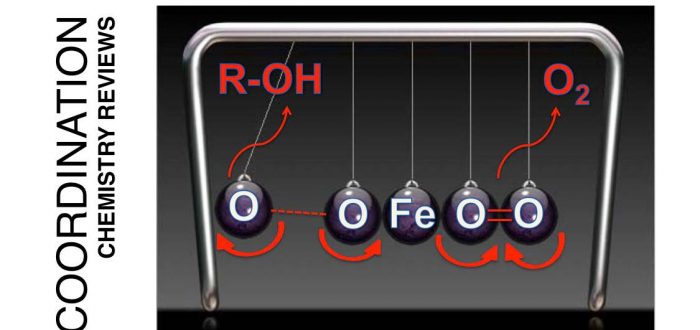
Lyses and formation of the O–O bond constitute key reactions in Biochemistry. A quite common feature of these reactions in biological systems is that they are catalyzed by first row transition metals, among which, iron, manganese and copper are prevalent. Their ability to support different oxidation states, especially high-valent metal-oxo species in the cases of iron and manganese, constitutes a key element to enable the red-ox processes associated with O–O bond formation/lysis. Synthetic bioinorganic chemistry has provided important contributions to understand O2 metabolism. Small molecule coordination complexes produced during the last two decades have been able to reproduce and facilitate understanding of the species that form during O–O bond forming and breaking reactions. The purpose of this account is to review and discuss selected examples where O–O formation and lysis at iron coordination complexes has been documented. The review is specially focused on reactions performed by iron complexes with aminopyridine ligands, where our research has been particularly focused. This review was published today in Coordination Chemistry Reviews (DOI 10.1016/j.ccr.2016.11.007):
I. Gamba, Z. Codolà, J. Lloret-Fillol, and M. Costas
“Making and breaking of the OO bond at iron complexes”
Coord. Chem. Rev. 2017, 334, 2-24
[abstract]
DOI: 10.1016/j.ccr.2016.11.007
- sec.iqcc@udg.edu
- +34 972 41 83 57