- sec.iqcc@udg.edu
- +34 972 41 83 57
Anna Roglans
Transition Metals in Organic Synthesis.
Contact info:
Dr. Anna Roglans
anna.roglans@udg.edu
Tel. (+34) 972 41 82 75
Website

Selected publications
Dr. Anna Roglans
Research overview
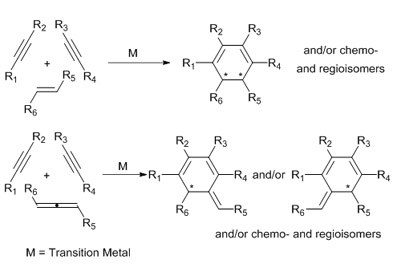
The use of alkenes and allenes as highly versatile substrates in cyclizations to create chiral polycyclic compounds
The involvement of alkenes and allenes in transition-metal calalyzed [2+2+2] cycloaddition reactions has the advantage of allowing for the eventual introduction of stereogenic centres in the newly formed six-membered ring and of affording complex polycyclic scaffolds. Our work is focused on the use of properly substituted alkenes, which are more reluctant to react than alkynes, that allow the synthesis of chiral cyclohexadienes and their further functionalization by Diels-Alder reaction. We are also investigating the use of allenes, unsaturated substrates that are more reactive than alkenes and that deliver cycloadducts with an exocyclic double bond that can subsequently be manipulated. One of the main aims in our research is to control the chemoselectivity and regioselectivity of these processes by determining the factors that govern them both experimentally and theoretically.
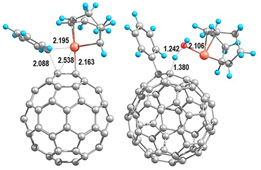
The functionalisation of fullerenes by cycloaddition reactions to provide a variety of potential molecular materials for biological, optical and electronic devices
Fullerenes are a class of molecule made up of carbon atoms with an unusual hybridization (sp2,3) exhibiting a chemical reactivity similar to that of electron-deficient olefins. [60]Fullerene, in particular, has received considerable attention due to its interesting spherical structures and unique physical and chemical properties. The functionalization of [60]fullerene with different functional groups provides a variety of potential molecular materials for biological, optical, and electronic devices. Our work is focused on exploring the versatility of rhodium complexes in the cycloaddition of pi-unsaturations to C60 as a tool to functionalize these molecules in a straightforward manner.
Mechanistic studies of transition-metal catalyzed reactions through electrospray ionization mass spectrometry (ESI-MS)
In order to effectively control and improve the catalytic reactions, it is necessary to understand the mechanistic details of the process. One of the difficulties faced in these investigations is that the low concentrations and transient nature of most of the intermediates involved make their identification difficult. Therefore, techniques that allow the direct monitoring of these reactive intermediates are of great interest. One of our research lines is to use electrospray ionization mass spectrometry (ESI-MS) and subsequent MS/MS analysis to obtain detailed data by trapping and identifying short-lived intermediates in transition-metal catalyzed [2+2+2] cycloaddition reactions. In addition to observing the molecular mass of ions for many reactive intermediates, this technique reveals structural information through characteristic fragmentation patterns by collision-induced dissociation. All these data help us to more fully understand the course of cycloadditions and allow us to improve these reactions.

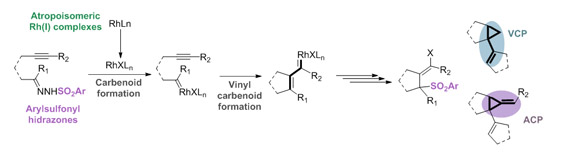
Methodological studies of cyclizations through rhodium carbenoid intermediates generated from tosylhydrazones
Research line led by Dr. Anna Pla-Quintana
The vast majority of syntheses rely on step-by-step transformations. This is an inefficient, time-consuming strategy that would be greatly improved by the implementation of effective cascade processes. Of the many intermediates that can initiate or participate in cascade processes, metal-carbenoids are an excellent option due to their versatility and high reactivity. Rhodium(II) dimers have emerged as the most versatile class of complexes for single-step carbenoid mediated reactions. The transition-metal carbenoids that are required for these reactions are mostly prepared from nitrogen extrusion of diazo compounds that have non-negligible security and toxicity risks. Furthermore, the methodology is limited just to carbenoids with certain electronic properties. This research line is aimed at preparing rhodium carbenoids making use of rhodium(I) complexes to decompose arylsulfonylhydrazones, a carbenoid source that is much safer and easier to handle and which is amenable to all types of carbenoids. Rhodium(I) complexes with atropoisomeric ligands are chosen to exploit their versatility and stereoselectivity in cycloaddition reactions, nucleophilic substitutions and rearrangements. This combination is selected to trigger cascade reactions that allow the straightforward enantioselective synthesis of (poly)cyclic compounds from readily available linear starting materials.
People
Principal Investigator
Staff and Postdocs
PhD and MACMoM students
Funding
MINECO Projects. Proyectos I+D.
Researcher: Dr Miquel Solà & Dr Anna Roglans
Reference: CTQ2014-54306-P
Funding: 196.020 €
Periode: 2015 – 2017
News
First Science Slam IQCC within Science Week
The Institute of Computational Chemistry and Catalysis of the University of Girona organizes the 1st Science
Review on allenes featured on the cover of Chem. Soc. Rev.
A tutorial review on the use of allenes in transition metal catalyzed cyclodadditions
Second edition of Science Slam IQCC, November 24, 12.00h
In 2015, the IQCC organized for the first time the Science Slam, which
Congratulations to all (previous) members of IQCC
At the beginning of August, the Nature Publishing Group sent out a mail




