- sec.iqcc@udg.edu
- +34 972 41 83 57
Solà, Miquel
Catalysis and Aromaticity
Contact info:
Dr. Miquel Solà
miquel.sola@udg.edu
Tel. (+34) 972 41 89 12
Website

Selected publications
Jordi Poater, Pascal Vermeeren, Trevor A. Hamlin, F. Matthias Bickelhaupt, Miquel Solà
On the existence of collective interactions reinforcing the metal-ligand bond in organometallic compounds
Nat Commun, 2023, 14, –
DOI: 10.1038/s41467-023-39498-y
Fangxiang Sun, Shuaimin Tan, Hou-Ji Cao, Chang-sheng Lu, Deshuang Tu, Jordi Poater, Miquel Solà, Hong Yan
Facile Construction of New Hybrid Conjugation via Boron Cage Extension
J. Am. Chem. Soc, 2023, 145, 3577-3587
DOI: 10.1021/jacs.2c12526
Miquel Solà
Aromaticity rules
Nat. Chem., 2022, 14, 585-590
DOI: 10.1038/s41557-022-00961-w
Ouissam El Bakouri, Dariusz W. Szczepanik, Kjell Jorner, Rabia Ayub, Patrick Bultinck, Miquel Solà, Henrik Ottosson
Three-Dimensional Fully π-Conjugated Macrocycles: When 3D-Aromatic and When 2D-Aromatic-in-3D?
J. Am. Chem. Soc., 2022, 144, 8560-8576
DOI: 10.1021/jacs.1c13478
Simon Zank, JesúsManuel Fernández-García, Anton Stasyuk, Alexander Voityuk, Marcel Krug, Miquel Solà, Dirk Guldi, Nazario Martin
Initiating Electron Transfer in Doubly‐curved Nanographene Upon Supramolecular Complexation of C60
Angew. Chem. Int. Ed., 2022, 61, 1433-7851
DOI: 10.1002/anie.202112834
Dr. Miquel Solà
Research overview
Aromaticity of classical organic aromatic and heteroaromatic compounds
Aromatic species have rings or closed structures characterized by high electronic delocalization. With this in mind, our group has been pioneering in analyzing the aromaticity of classical aromatic and heteroaromatic compounds through electronic-based aromaticity measures. We have developed new aromaticity indices like PDI, FLU, INB and ING for such purpose, which perfectly complement others like the geometrical HOMA or the magnetic NICS. More recently, we have analyzed these heteroaromatic compounds (with especial emphasis of the corresponding isomerization energies) by means of Morokuma-like energy decomposition analysis and the turn-upside-down approach.
Metalloaromaticity
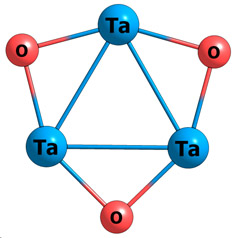
In the last decades the study of aromaticity has experienced an enormous progress. The new discoveries include species such as the metallabenzenes, heterometallabenzenes, metallabenzynes, metallabenzenoids, metallacyclopentadienes, metallacyclobutadienes and all-metal and semimetal clusters. These new molecules, which are potentially useful for certain purposes as specific and very efficient catalysts, molecular electronic devices, molecular magnets, drugs, and other as yet unimagined applications, have brought a complete revolution in the field. At variance with the classical aromatic organic molecules that possess only ?-electron delocalization, aromaticity in these new species is much more complex. These compounds have ?-, ?-, ?- and ?-electron delocalization. In addition, they can combine different types of aromaticity thus giving rise to double or triple aromaticity, the so-called multifold aromaticity. The new molecules can also have conflicting aromaticity, i.e., they can be aromatic in one component and antiaromatic in another. Moreover, most of the old indicators are not valid to discuss the complex aromaticity of these novel compounds. The analysis of metalloaromaticity in our group follows two main directions. First, we discuss the metalloaromaticity and the molecular properties of metallacycles and all-metal and semimetal clusters. And second, we aim to find reliable measures of aromaticity in these systems. In particular the use of multicenter electronic delocalization indices is advocated because they help to detect the different types of aromaticity and provide reasonable qualitative orderings of aromaticity.
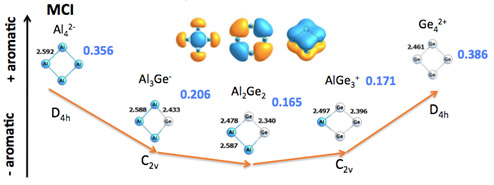
New Methods and Tools to Quantify Aromaticity in Molecules
Aromaticity is a concept of capital importance in chemistry: it governs chemical reactions, attributes extra stabilization to molecular structures and assigns exalted magnetic properties. Unfortunately, there is not a unique way to account for aromaticity and, as a result, many indices accounting for different manifestations of aromaticity are available in the literature. Our group has actively worked in the construction of new electronic aromaticity indices departing from the fact that aromaticity is related to cyclic electronic delocalization. For such, we have suggested several new measures, including PDI, FLU, INB and ING indices. Interestingly, the last two use the so-called multicenter indices, which have been proven to perform the best in different chemical enviroments (from organic to all-metal aromatic compounds), together with the magnetic-based nucleus independent chemical shifts (NICS).
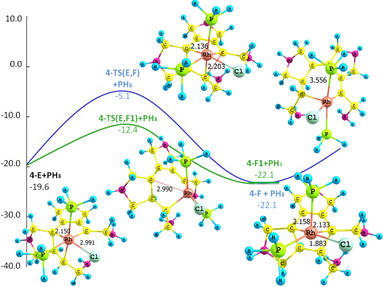
Organometallic chemistry: reaction mechanisms and computations.
In this line of research we carry out extensive mechanistic studies, i.e. we determine the reaction path followed from reactants to products, with localization of intermediates and transition states in organometallic and bioinorganic reactions. Most of the mechanisms investigated in this research line yield products of special interest either at large-scale manufacturing or because of their properties as powerful drugs, antibiotics or anticancer agents. Since in these cases obtaining the desired product (or, on the contrary, of an undesired by-product) can represent a gain (or a loss) of many million dollars every day, and since the final result is mainly governed by the path followed by the reaction, there is a great incentive to improve the understanding of the details of the operative mechanism of a given chemical process.
Fullerene Chemistry and Electron Transfer in Biomolecules and Materials
Fullerene-based architectures are being intensively studied as electron acceptor materials in the construction of plastic photovoltaic cell devices because of their unique structural and electron acceptor properties. Organic solar cells containing fullerenes have already been commercialized but the current power conversion efficiency (PCE) achieved is less than 10%. Most of the fullerene-based photovoltaic cells are based on the bulk heterojunction concept, where a donor is blended with a fullerene-based derivative that acts as acceptor. Possible alternatives to improve the current efficiencies in such organic solar cells involve the covalent linkage of the donor molecule with the fullerene surface. The covalent functionalization of the donor molecule to fullerenes and EMFs is mainly achieved via cycloaddition reactions, principally through Diels-Alder (DA), 1,3-dipolar (Prato) and nucleophilic [2+1] Bingel-Hirsch (BH) cycloaddition. In this project, we aim to computationally design new donor-acceptor conjugates with the ability to efficiently generate long-live charge separated states with lifetimes comparable to those observed in natural photosynthetic systems. To that aim, we explore the exohedral reactivity of several donor-acceptor systems based on empty fullerene cages and on EMFs to investigate the thermal stability of the adducts, the effect of the donor position with respect to the fullerene cages, and the ability to form photoinduced charge separated states.
People
Principal Investigator
Staff and Postdocs

Albert Artigas
Postdoc (BP)
Supervisor:- M. Solà
- A. Pla-Quintana
- A. Roglans

Alexander Voityuk
ICREA Research Professor
Emeritus - Collaborator
Jordi Mestres
Full Professor
Supervisor:- M. Sola

Nil Roig
Postdoc
Supervisor:- A. Pla-Quintana
- M. Solà

Ouissam El Bakouri
Postdoc (ONCE)
Supervisor:- M. Solà
- A. Poater

Roger Monreal
Postdoc
Supervisor:- A. Pla-Quintana
- A. Poater
PhD and MACMoM students

Iqra Sarfraz
PhD student (IF-UdG)
Supervisor:- M. Solà

Joan Cabot
PhD student (DI)
Supervisor:- J. Mestres
- M. Solà
Jordi Busoms
PhD student
Supervisor:- J. Mestres
- M. Solà

Louis Van Nyvel
PhD Student
Supervisor:- M. Solà

Maria Cabrero
PhD student (FPI)
Supervisor:- M. Solà

Maria Martínez
PhD Student (DI)
Supervisor:- J. Mestres
- M. Solà

Mariona Isern
PhD student
Supervisor:- J. Mestres
- M. Solà
Nikola Panajotovikj
PhD student
Supervisor:- J. Mestres
- M. Solà

Omkar Charapale
PhD student (FI)
Supervisor:- M. Solà

Raquel Parrondo
PhD student
Supervisor:- J. Mestres
- M. Solà

Núria Alsina
PhD Student (FI-SDUR)
Supervisor:- S. Simon
- M. Rodríguez
- M. Iglesias
Funding
ICREA Academia
Researcher: Prof. Miquel Solà
Reference: 2024 ICREA 00058
Funding: 200.000 €
Period: 01/01/2025 – 31/12/2029
MCIU Proyectos I+D. Proyectos I+D
Researcher: Dr. Jordi Mestres
Reference: PID2023-153094OB-I00
Funding: 87.500 €
Period: 01/09/2024 – 31/12/2027
Researcher: Prof. Miquel Solà
Reference: PID2023-147424NB-I00
Funding: 237.500 €
Period: 01/09/2024 – 31/12/2027
UdG
Researcher: Jordi Mestres (IQCC, UdG) and Xavier Jalencas (Chemotargets)
Reference: 2024 DI 00105
Funding: 37.800 €
Period: 2025 – 2028
MICIU. Redes temáticas
Researcher: Dr. Agustí Lledó and Prof. Miquel Solà
Reference: RED2024-154178-T
Funding: 28.000 €
Period: 2025 – 2027
Marie Skłodowska-Curie
Project: #NitRecerCat2425: La Nit Europea de la Recerca
Researcher: Dr. Sílvia Simon
Reference: HORIZON-MSCA-2024-101162003
Funding: 300.000 €
Period: 01/07/2024 – 30/06/2026
AGAUR Project. Beatriu de Pinós
Researcher: Dr. Albert Artigas (M. Solà)
Reference: 2023 BP 00033
Funding: 152.348 €
Period: 2025-2028
Doctorats industrials
Researcher: –
Reference: 2024 DI 00015
Funding: 37.800 €
Period: –
Researcher: Prof. Miquel Solà
Funding: 24.500 €
Period: 2024-2026
Collaborations
Mercedes Alonso – Vrije Universiteit Burssels (Belgium), collaboration with Miquel Solà.
Ricardo Pino-Rios – Universidad Arturo Prat (Chile ), collaboration with Miquel Solà.
Álvaro Muñoz-Castro – Universidad San Sebastián (Chile ), collaboration with Miquel Solà.
Jun Zhu – University of Xiamen (China), collaboration with Miquel Solà.
Hong Yang – Nanjing University (China), collaboration with Miquel Solà.
José Oscar Carlos Jiménez-Halla – Univ. Guanajuato (México), collaboration with Miquel Solà.
J. Oscar C. Jiménez-Halla – Universidad de Guanajuato (México), collaboration with Miquel Solà.
F. Matthias Bickelhaupt – Vrije Universiteit Amsterdam (Netherlands), collaboration with Miquel Solà.
Juan Pablo Martínez – University of Warsaw (Poland), collaboration with Miquel Solà.
Tadeuz M. Krygowski – University of Warsaw (Poland), collaboration with Miquel Solà.
Halina Szatylowicz – Warsaw University of Technology (Poland), collaboration with Miquel Solà.
Dariusz W. Szczepanik – Jagiellonian University (Poland ), collaboration with Miquel Solà.
Slavko Radenkovic – Kragujevac University (Serbia), collaboration with Miquel Solà.
Francesc Teixidor – Institut de Ciència dels Materials (ICMAB-CSIC) (Spain), collaboration with Miquel Solà.
Clara Viñas – Institut de Ciència dels Materials (ICMAB-CSIC) (Spain), collaboration with Miquel Solà.
Jordi Poater – Univ. Barcelona (Spain), collaboration with Miquel Solà.
Nazario Martín – Univ. Complutense de Madrid (Spain), collaboration with Miquel Solà.
Henrik Ottosson – Uppsala University (Sweden), collaboration with Miquel Solà.
News
PhD defense Roger Monreal Corona
On 14th of January will be the defense of the doctoral thesis of Roger
Two former IQCC researchers awarded Ramón y Cajal contracts
Two former members of the IQCC were obtained the Ramón y Cajal contract with
Cover Chemical Science on Enhancing the Statistical Probability Factor in Triplet-Triplet Annihiltaion Photon Upconversion via TIPS Functionalization
The Chem. Sci. journal features on its front cover of the November 2025 issue
Seven IQCC researchers have been included in Stanford University global list of 2% top scientists
The standardized citation metric analysis published by Ioaniddis (popularized as Stanford University global list








