He works in the field of theoretical (bio)inorganic and supramolecular chemistry, and works on transition-metal complexes, metalloproteins, enzymes, and DNA. The effect of (transition) metal ions on reactivity, selectivity and chemical bonding is one of the main topics in these studies. The development of computational tools for these studies is an important ingredient, to which he has contributed largely both with his own software (QUILD, DRF90) as in contributions in general purpose software (ADF/AMS, CFOUR, NWCHEM).
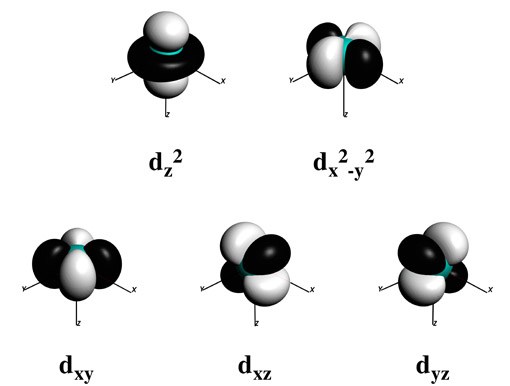
Spin states of transition-metal complexes
It has long been recognized that metal spin states play a central role in the reactivity of important biomolecules and in inorganic chemistry catalysis. Molecules with different numbers of unpaired electrons, hence with different spin states, have distinct geometric structures, energetic properties and reactivity. Elucidating the role and effect of different spin states on the properties of a system is presently one of the most challenging endeavors both from an experimental and theoretical point-of-view. This is in particular true for reaction mechanisms where the spin-state and/or oxidation state of a metal can change during the reaction. It has been described extensively in the recent Wiley book on “Spin states in Biochemistry and inorganic chemistry: Influence on Structure and Reactivity” (Eds: M. Swart, M. Costas).

Transition-metal reactivity studies
The reaction mechanism of chemical processes with transition-metals can (and often does) involve a switching from one spin-state to another during the reaction. This allows to overcome the low activity of molecules like dioxygen (in its triplet state), and facilitates reactions that would otherwise be spin-forbidden. Moreover, given the wide range of mechanistic possibilities available with the presence of transition-metals, often a multitude of reaction mechanisms has to be considered. In collaboration with experimental groups we are carrying out computational chemistry experiments to characterize intermediates and transition structures, to determine the most favorable reaction path.
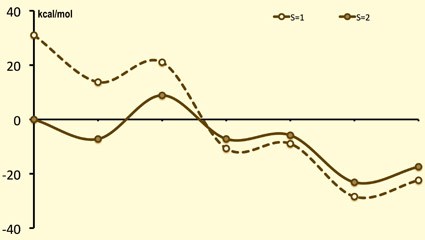
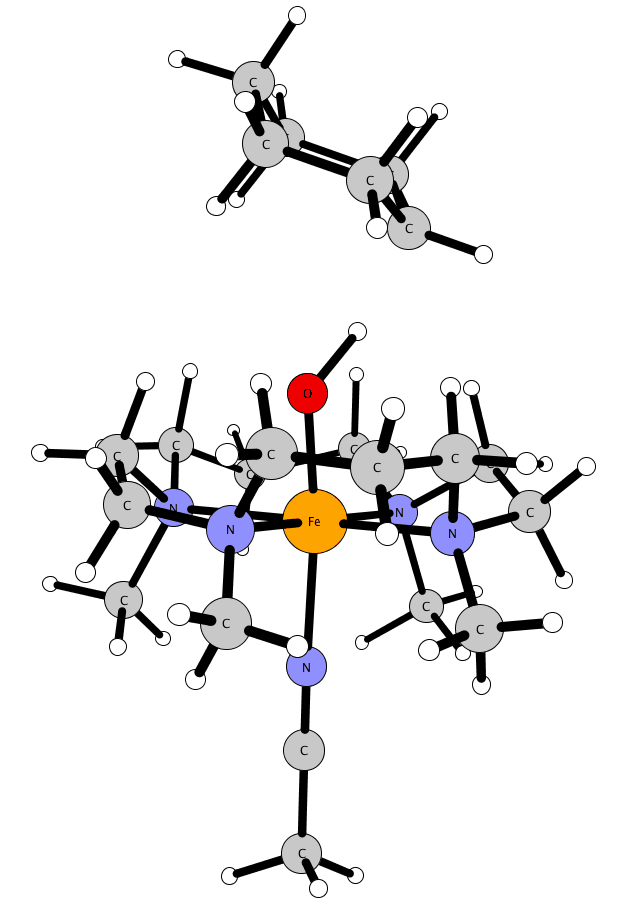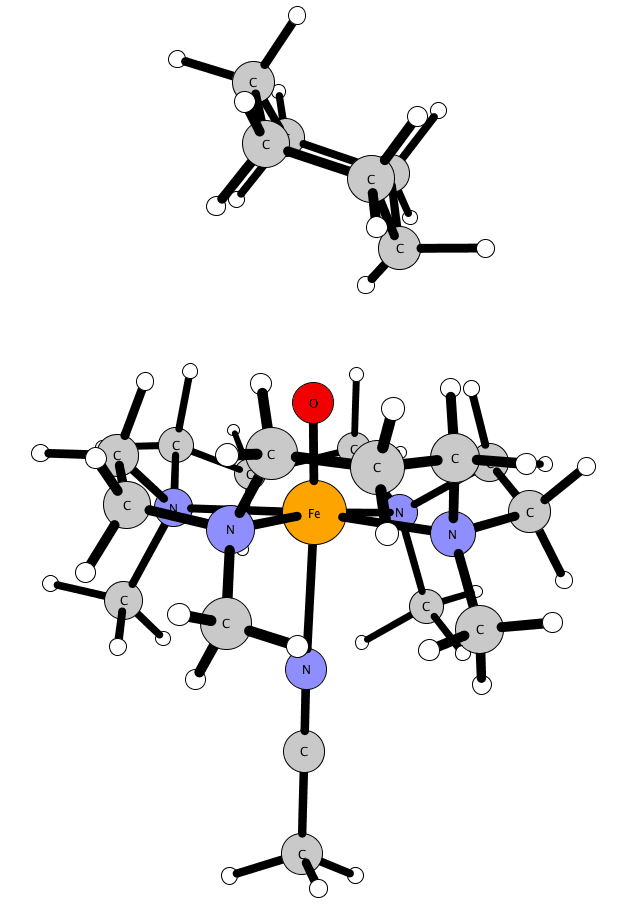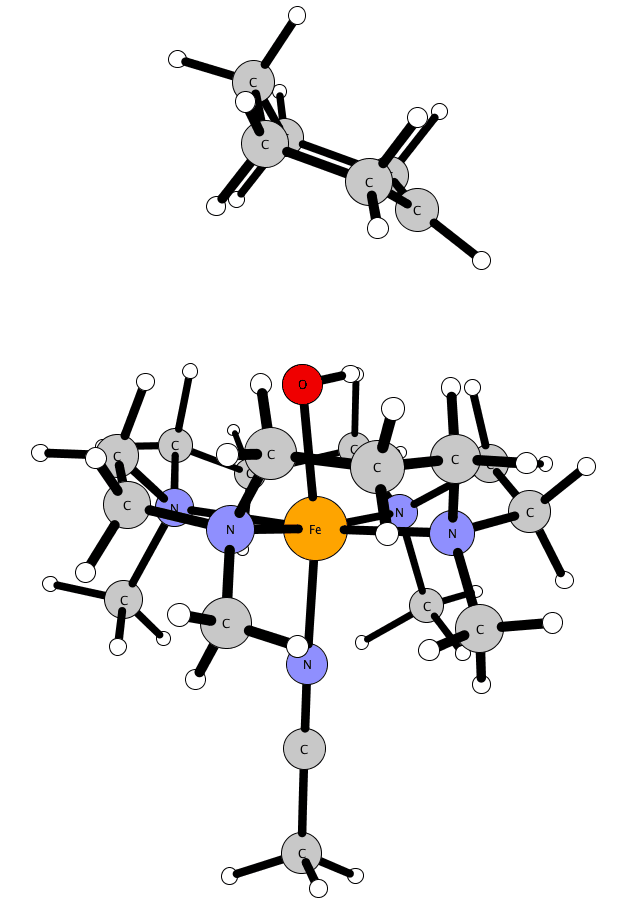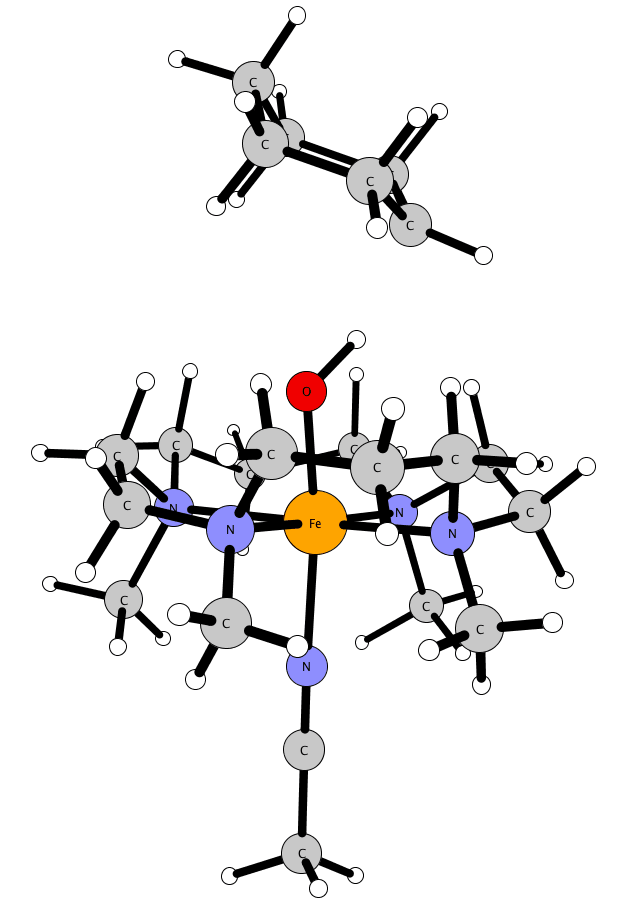
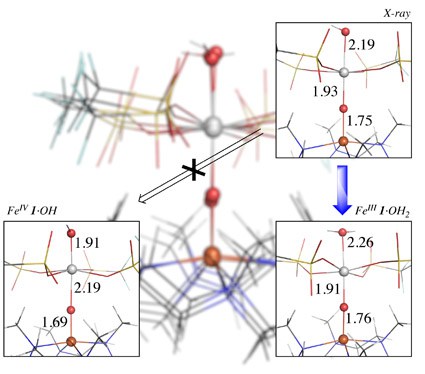
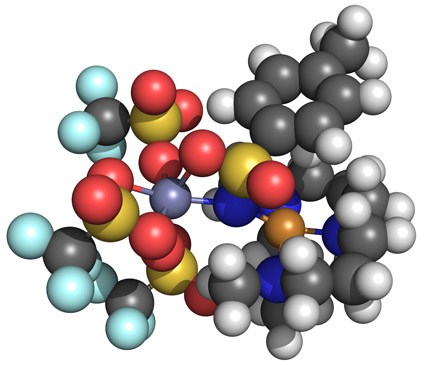
Lewis acids: stabilization of vulnerable species and reactivity enhancement
In 2010 a ground-breaking experimental study by Fukuzumi and Nam was published in Nature Chemistry where they showed that adding Sc(OTf)3 to a FeIV-oxo complex (with the TMC ligand) has unprecedented consequences: (i) the axial ligand of iron is removed; (ii) scandium picks up a fourth triflate and an additional fifth axial ligand; (iii) the methyl groups of the TMC-ligand flip upwards (from anti towards oxo, to syn); and (iv) this interaction facilitates a two-electron reduction by ferrocene, instead of the one-electron reduction without the Lewis acid. This has opened a whole new research area where reactive species can be stabilized, or reactivity can be drastically improved. The effect of the Lewis acids on the stability, reactivity and spectroscopic properties is poorly understood, and is being studied here in collaboration with experimental groups.
Development of computational research tools
In order to be able to carry out the spinning around in transition-metal chemistry, new research tools have been developed such as the QUILD program with improved optimization and transition-search routines, and new density functionals (S12g, SSB-D) that work well for both spin states and weak interactions. The advantage of these tools is that they can be applied straightforwardly also to other systems such as DNA, SN2-reactions, NMR chemical shifts and the like