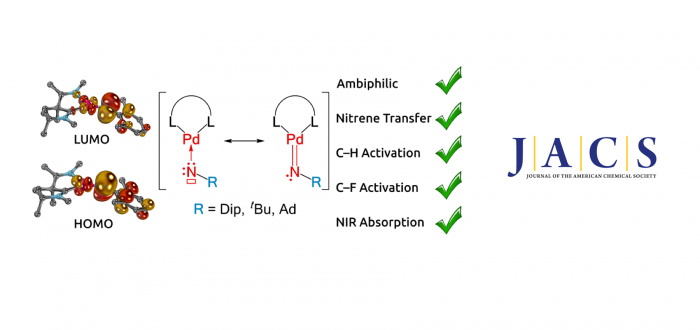
Triplet-nitrene complexes of the late transition metals are isolable and key intermediates in catalysis. However, singlet-nitrene ligands remain elusive. In this article, we reported three such palladium terminal imido complexes with singlet ground states. Combining UV-vis-NIR electronic spectroscopy, which revealed broad bands up to 1400 nm, with high-level quantum-chemical computations (DLPNO-STEOM-CCSD, CASSCF/NEVPT2 and KS-DFT coupled to EOS analysis) suggest significant palladium(0) singlet-nitrene character. Although the aliphatic nitrene complexes proved to be too reactive for isolation in analytically pure form as a result of olefin elimination, the aryl congener could be cha¬rac¬terized by SC-XRD, elemental analysis, IR-, NMR spectroscopy, and HRMS. The complexes’ distinguished ambiphilicity corroborates strong nitrene character. It allows them to activate hexafluorobenzene, triphenylphosphine, and pinacol borane, catalytically dehydrogenate cyclohexene, and to aminate ethylene via nitrene transfer at or below room temperature.
This work has been performed by the Munz group at Saarland University in collaboration with one of our IQCC members, Martí Gimferrer, who performed the computational analysis of the metal–nitrene bond. This work was recently published in Journal of the American Chemical Society:
A. Grünwald, B. Goswami, K. Breitwieser, B. Morgenstern, M. Gimferrer, F. W. Heinemann, D. M. Momper, C. W. M. Kay, and D. Munz
“Palladium Terminal Imido Complexes with Nitrene Character”
J. Am. Chem. Soc. 2022, ASAP
DOI: 10.1021/jacs.2c02818
Girona, May 28, 2022
For more info: gestor.iqcc@gmail.com