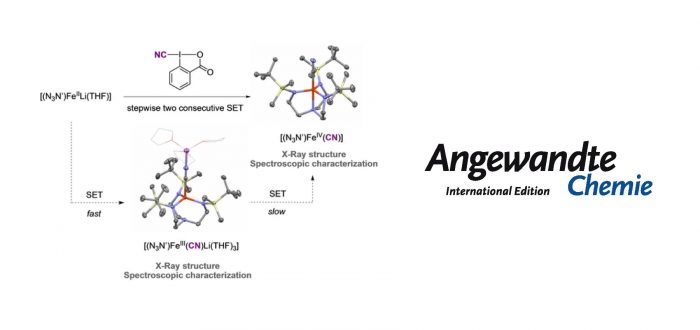
Cyano lambda3-iodanes react with iron(II) and iron(III) complexes as cyano-transfer one-electron oxidants. This approach enables the straightforward synthesis of iron(III) and iron(IV) cyanide complexes, which have been thoroughly characterized by spectroscopic methods, including X-ray diffraction, EPR, Mössbauer and NMR spectroscopies and computational methods.
We disclose a new reactivity mode for electrophilic cyano lambda3-iodanes as group transfer one-electron oxidants to synthesize FeIII and FeIV cyanide complexes. The inherent thermal instability of high-valent Fe(IV) compounds without pi-donor ligands (such as oxido (O2–), imido (RN2–) or nitrido (N3–)) makes their isolation and structural characterization a very challenging task. We report the synthesis of an FeIV cyanide complex [(N3N’)FeCN] (4) by two consecutive single electron transfer (SET) processes from Fe(II) precursor [(N3N’)FeLi(THF)] (1) with cyanobenziodoxolone (CBX). The Fe(IV) complex can also be prepared by reaction of [(N3N’)FeIII] (3) with CBX. In contrast, the oxidation of Fe(II) with 1-cyano-3,3-dimethyl-3-(1H)-1,2-benziodoxole (CDBX) enables the preparation of Fe(III) cyanide complex [(N3N’)FeIII(CN)(Li)(THF)3] (2-LiTHF). Complexes 4 and 2-LiTHF have been structurally characterized by single crystal X-ray diffraction and their electronic structure has been examined by Mössbauer, EPR spectroscopy, and computational analyses.
The paper is a collaboration of former member Alicia Casitas and Marcel Swart and co-workers, and was recently published in Angewandte Chemie-International Edition:
C. Souilah, S.A.V. Jannuzzi, D. Demirbas, S. Ivlev, M. Swart, S. DeBeer and A. Casitas
“Synthesis of Fe(III) and Fe(IV) Cyanide Complexes Using Hypervalent Iodine Reagents as Cyano-Transfer One-Electron Oxidants”
Angew. Chem. Int. Ed. 2022, ASAP
DOI: 10.1002/anie.202201699
Girona, March 18, 2022
For more info: gestor.iqcc@gmail.com