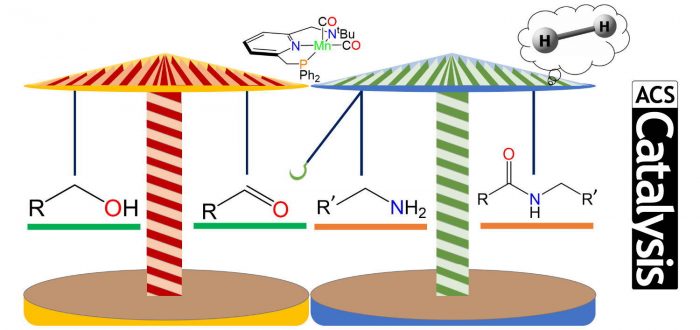
Density functional theory calculations unveil the mechanism of the original example of a base-metal-catalyzed synthesis of amides from alcohols and amines, generating H2 as a subproduct. Instead of a convoluted mechanism with the implication of dissociation of some ligand of the manganese catalyst, our DFT calculations describe a facile protocol, where the catalyst only produces aldehydes from alcohols. Once formaldehyde is formed from methanol, it reacts with the amine to form a second alcohol that undergoes the same procedure as methanol and creates the desired amide through a double-carrousel mechanism. This reaction is eminently sustainable in different ways: it uses alcohols and generates molecular hydrogen, without releasing CO2, but the interesting formation of amides. In addition, to speed up the process the reaction has a manganese catalyst, with manganese being one of the most abundant metals in the Earth’s layer.
The paper by the teams of Miquel Solà and Albert Poater has recently been published in ACS Catalysis:
J.A. Luque-Urrutia, T. Pèlachs, M. Solà, and A. Poater
“Double-Carrousel Mechanism for Mn-Catalyzed Dehydrogenative Amide Synthesis from Alcohols and Amines”
ACS Catal. 2021, 11, 6155-6161
DOI: 10.1021/acscatal.1c00693
Girona, May 8, 2021
For more information: gestor.iqcc@gmail.com