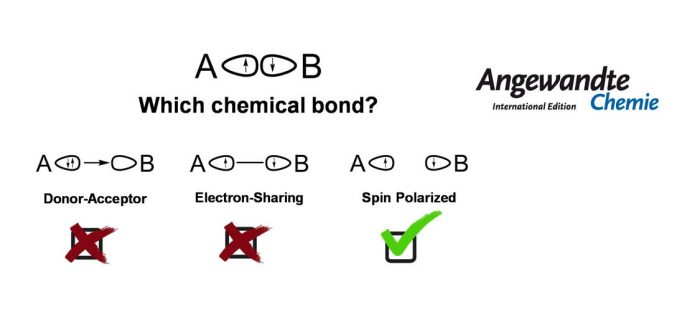
What is a chemical bond? The chemical bond is a central concept for describing and predicting molecular structure and reactivity. Since a chemical bond does not have an operator in quantum mechanics, there is no exclusive approach to describe it unequivocally. A fundamental approach towards understanding its properties consists in classifying the electron-pair interactions between atoms or fragments. There are two well-established classes of bonding interactions, according to the origin of the electron-pair. When each fragment contributes with one electron, the bonding is described as a covalent electron-sharing bond. When both electrons are contributed by one of the fragments, the interaction is interpreted as a dative or donor-acceptor bond. However, for systems with marked multi-reference character, this binary categorization might not be precise enough to render the bonding properties.
In the current contribution, a third scenario is discussed in detail, namely the case of spin polarized bonds. An important insight into chemical bonding analysis has been provided by simple model, based on the bond order and local spin indicators, which discriminates between all three bonding situations. The recently discussed case of the exotic NaBH3– cluster appears to be best described as a spin-polarized diradicaloid.
This paper is the result of a collaboration between the groups of Dr. Pedro Salvador, Dr. Inés Corral (Autonomic University of Madrid), and Dr. Diego Andrada (University of Saarland); the three groups have combined their expertise in chemical bonding from different perspective, namely, topology, molecular orbitals and multi-reference calculations. The paper was published recently in Angewandte Chemie:
D.M. Andrada, P. Salvador, E. Vos, and I. Corral
“Beyond the Classical Electron?Sharing and Dative Bond Picture: The Case of Spin?Polarized Bond”
Angew. Chem. Int. Ed. 2020, ASAP
DOI: 10.1002/anie.202010948
Girona, September 26, 2020
More information: gestor.iqcc@gmail.com