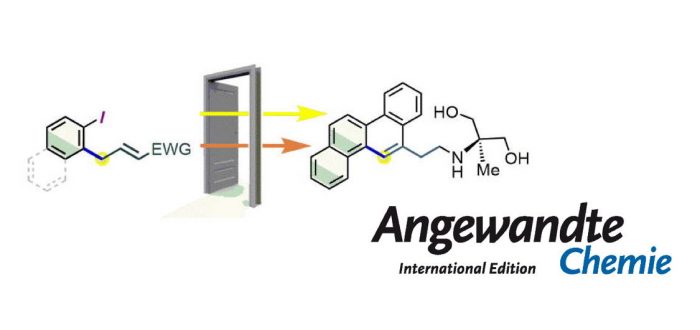
A metal-free C-H allylation strategy is described to access diverse functionalized ortho-allyl-iodoarenes. The method employs hypervalent (diacetoxy)iodoarenes and proceeds through the iodane-guided “iodonio-Claisen” allyl transfer. The use of allylsilanes bearing electron-withdrawing functional groups unlocks the functionalization of a broad range of substrates, including electron-neutral and electron-poor rings. The resulting ortho-allylated iodoarenes are versatile building blocks, with examples of downstream transformation including a concise synthesis of the experimental antimitotic core of Dosabulin. DFT calculations shed additional light on the reaction mechanism, with notable aspects including the aromatic character of the transition-state structure for the [3,3] sigmatropic rearrangement, as well as the highly stereoconvergent nature of the trans-product formation.
This paper is the result of a joint collaboration with the groups of Prof. A. Shafir of the Institute for Advanced Chemistry of Catalonia, Prof. A. B. Cuenca of the Químics de Sarrià, Prof. S. Lethu of the Institute of Chemical Research of Catalonia, and Prof. J. Zhu from Xiamen University; our group has performed the DFT mechanistic study with a great work by Dandan Chen. The paper was published recently in Angewandte Chemie:
W.W. Chen, A. Cunillera, D. Chen, S. Lethu, A. López de Moragas, J. Zhu, M. Solà, A.B. Cuenca, and A. Shafir
“Iodane-Guided ortho C-H Allylation”
Angew. Chem. Int. Ed. 2020, ASAP-
DOI: 10.1002/anie.202009369
Girona, September 3, 2020
More information: gestor.iqcc@gmail.com