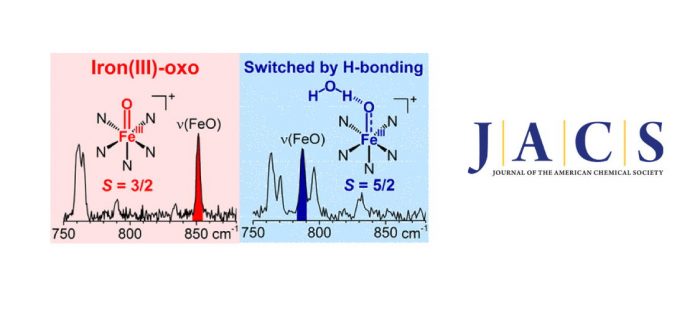
Today, Miquel Costas and co-workers reported in JACS on the trapping of Fe(III)=oxo species. Terminal non-heme iron(IV)-oxo compounds are among the most powerful and best studied oxidants of strong C-H bonds. In contrast to the increasing number of such complexes (>80 thus far), corresponding one-electron-reduced derivatives are much rarer and presumably less stable, and only two iron(III)-oxo complexes have been characterized to date, both of which are stabilized by hydrogen-bonding interactions. Herein we have employed gas-phase techniques to generate and identify a series of terminal iron(III)-oxo complexes, all without built-in hydrogen bonding. Some of these complexes exhibit ?70 cm-1 decrease in ?(Fe-O) frequencies expected for a half-order decrease in bond order upon one-electron reduction to an S = 5/2 center, while others have ?(Fe-O) frequencies essentially unchanged from those of their parent iron(IV)-oxo complexes. The latter result suggests that the added electron does not occupy a d orbital with Fe?O antibonding character, requiring an S = 3/2 spin assignment for the nascent FeIII-O– species. In the latter cases, water is found to hydrogen bond to the FeIII-O– unit, resulting in a change from quartet to sextet spin state. Reactivity studies also demonstrate the extraordinary basicity of these iron(III)-oxo complexes. Our observations show that metal-oxo species at the boundary of the “Oxo Wall” are accessible, and the data provide a lead to detect iron(III)-oxo intermediates in biological and biomimetic reactions.
E. Andris, R. Navrátil, J. Jašík, M. Puri, M. Costas, L. Que, and J. Roithová
“Trapping Iron(III)–Oxo Species at the Boundary of the “Oxo Wall”: Insights into the Nature of the Fe(III)–O Bond”
J. Am. Chem. Soc. 2018, 140, 14391-14400 [abstract]
DOI: 10.1021/jacs.8b08950